


This shows that cathode rays are made up of material particles.ģ)When an electric field is applied on the cathode rays, they are deflected towards the positive plate of the electric field. This shows that cathode rays travel in straight line.Ģ)If a light paddle wheel mounted on an axle is placed in their path, the wheel begins to rotate. This shows that some invisible rays are emitted from the cathode which passed through the holes of the anode and strike the wall.These rays were called as cathode rays.ġ)They produce a sharp Shadow of the solid object placed in their path. When high voltage of about 10000 volts is applied between the electrodes the following results are observed at different pressures :ġ) When the gas pressure inside is one atmosphere, no current flows between the electrodes.Ģ) When the pressure is reduced to about 10 millimetre ,the current starts flowing between the electrodes and coloured glow is observed ,the colour depending upon the nature of the gas taken.ģ) When the pressure is for the reduced to about 0.01 millimetre ,the glow between the electrodes disappear but the current continuous to flow and if a perforated anode is used, a faint greenish glow is observed on the glass walls behind the anode.
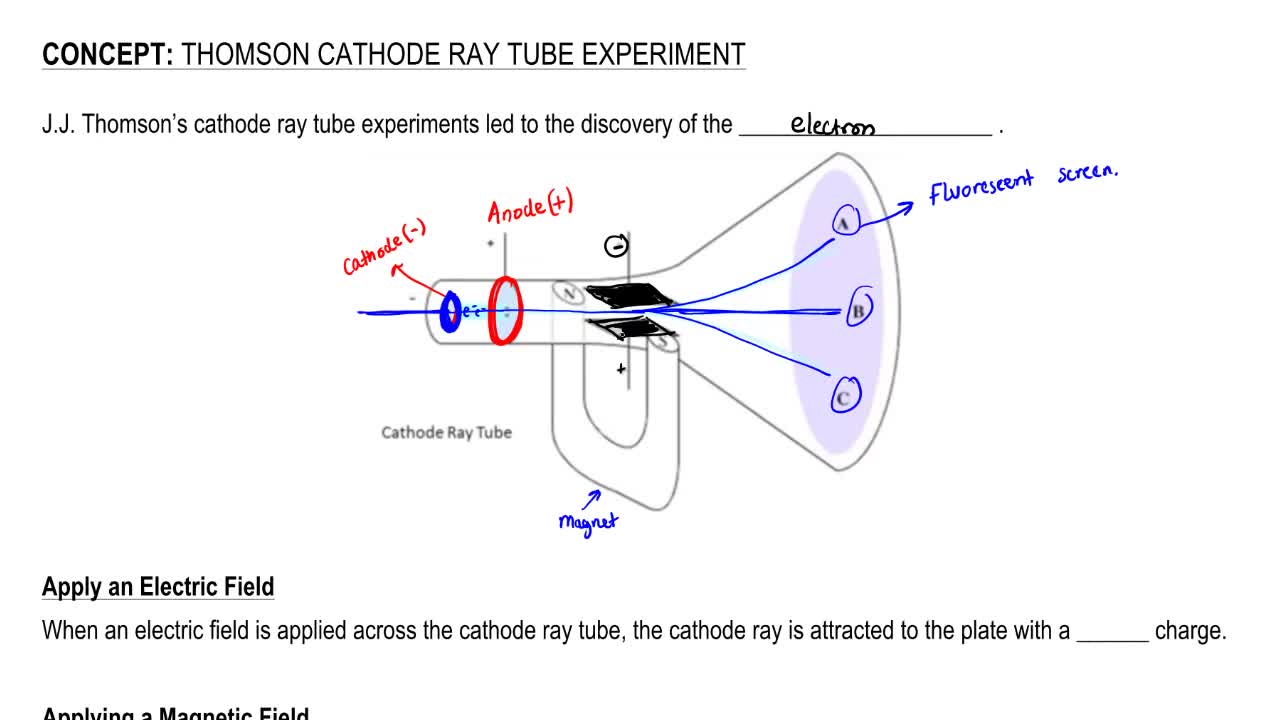
Measured masses and charges of the three elementary particles are given in the following table.For a neutral atom, number of proton=number of electron.Atomic mass = mass of protons + mass of neutron.The gold foil experiment was originally conducted by Hans Geiger (left) and Ernest Marsden (right) under the supervision of Ernest Rutherford at the University of Manchester. James Chadwick (on the right), a student of Rutherford won Nobel Prize in Physics in 1935 for discovery of neutron. Ernest Rutherford (left) was awarded Nobel Prize in Chemistry in 1908 for his work in radioactivity. Here the symbol  \(_Z^XA\) is used where Z = atomic number and X = atomic mass of the element A. Figure 7. Thomson measured the charge-by-mass-ratio (e/m) of cathode ray particle using deflection in both electric and magnetic field. Deflection of cathode rays towards positively charged plates Hence cathode ray consists of negatively charged particles. Applying electric field in the path of cathode ray deflects the ray towards positively charged plate. They are independent of the material composition of the cathode.ģ. The schematic of a cathode ray tube is given in figure 2. This ray is called cathode ray and the whole construction is called cathode ray tube. He detected that a stream of particle (ray) was coming out from the negatively charged electrode (cathode) to positively charged electrode (anode). Then he applied a high electrical voltage between two electrodes at either end of the tube. much of the air was pumped out of the tube. Thomson constructed a glass tube which was partially evacuated i.e. Thomson also won the Nobel Prize in 1937 for proving the wavelike properties of electron. Thomson won Nobel Prize in 1906 for discovering the elementary particle electron. Thomson in 1897 when he was studying the properties of cathode ray. Discovery of Electron Who discovered the electron?Įlectron was discovered by J.
